Khan Academy on a Stick
Substitution and elimination reactions
SN1, SN2, E1, E2, nucleophiles, nucleophilicity, basicity
-
 Naming Alkyl Halides
cc
Naming Alkyl Halides
ccNaming Alkyl Halides
-
 Sn2 Reactions
cc
Sn2 Reactions
ccSn2 Reactions
-
 Sn1 Reactions
cc
Sn1 Reactions
ccIntroduction to Sn1 reactions
-
 Steric hindrance
Steric hindrance
-
 Sn2 Stereochemistry
cc
Sn2 Stereochemistry
ccSn2 Stereochemistry
-
 Solvent Effects on Sn1 and Sn2 Reactions
cc
Solvent Effects on Sn1 and Sn2 Reactions
ccSolvent Effects on Sn1 and Sn2 Reactions
SN1 vs SN2
In this tutorial, Sal analyzes the differences between SN1 and SN2 reactions.
-
 Nucleophilicity (Nucleophile Strength)
cc
Nucleophilicity (Nucleophile Strength)
ccNucleophilicity (Nucleophile Strength)
-
 Nucleophilicity vs. Basicity
cc
Nucleophilicity vs. Basicity
ccNucleophilicity vs. Basicity . The difference between what it means to be a nucleophile and a base
Nucleophilicity and basicity
In this tutorial, Sal discusses the difference between nucleophilicity and basicity.
-
 E2 Reactions
cc
E2 Reactions
ccE2 Elimination Reactions
-
 E1 Reactions
cc
E1 Reactions
ccE1 Elimination Reactions
-
 Zaitsev's Rule
cc
Zaitsev's Rule
ccZaitsev's Rule for E2 and E1 reactions
Elimination reactions
In this tutorial, Sal explains the difference between an E1 and an E2 elimination reaction.
-
 Comparing E2 E1 Sn2 Sn1 Reactions
cc
Comparing E2 E1 Sn2 Sn1 Reactions
ccComparing E2 E1 Sn2 Sn1 Reactions
-
 E2 E1 Sn2 Sn1 Reactions Example 2
cc
E2 E1 Sn2 Sn1 Reactions Example 2
ccE2 E1 Sn2 Sn1 Reactions Example 2
-
 E2 E1 Sn2 Sn1 Reactions Example 3
cc
E2 E1 Sn2 Sn1 Reactions Example 3
ccE2 E1 Sn2 Sn1 Reactions Example 3
SN1/SN2/E1/E2
In this tutorial, Sal compares the differences between E2, E1, SN2, and SN1 reactions.
SN1 and SN2
In this tutorial, Jay covers the definitions of nucleophile/electrophile, The Schwartz Rules (may the Schwartz be with you!), and the differences between SN1 and SN2 reactions.
-
 E1 Elimination: mechanism
E1 Elimination: mechanism
-
 E1 Elimination: regioselectivity and stereoselectivity
E1 Elimination: regioselectivity and stereoselectivity
-
 carbocations and rearrangements
carbocations and rearrangements
-
 E1 Elimination: carbocation rearrangements
E1 Elimination: carbocation rearrangements
-
 E2 Elimination: mechanism
E2 Elimination: mechanism
-
 E2 Elimination: regioselectivity
E2 Elimination: regioselectivity
-
 E2 Elimination: stereoselectivity
E2 Elimination: stereoselectivity
-
 E2 Elimination: stereospecificity
E2 Elimination: stereospecificity
-
 E2 Elimination: substituted cyclohexanes
E2 Elimination: substituted cyclohexanes
E1 and E2 reactions
In this tutorial, Jay covers the E1 elimination mechanism, carbocation rearrangements, and the details of the E2 elimination reaction.
-
 nucleophilicity and basicity
nucleophilicity and basicity
Correction: I forgot to draw a negative charge next to the first iodide anion.
-
 SN1 SN2 E1 E2 reactions: primary and tertiary alkyl halides
SN1 SN2 E1 E2 reactions: primary and tertiary alkyl halides
-
 SN1 SN2 E1 E2 reactions: secondary alkyl halides
SN1 SN2 E1 E2 reactions: secondary alkyl halides
SN1/SN2/E1/E2
In this tutorial, Jay discusses the strength of a nucleophile and the differences between SN1, SN2, E1, and E2 reactions.
 nucleophile/electrophile and The Schwartz Rules
nucleophile/electrophile and The Schwartz Rules alkyl halide nomenclature
alkyl halide nomenclature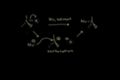 SN1 reaction: mechanism
SN1 reaction: mechanism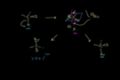 SN1 reaction: stereochemistry
SN1 reaction: stereochemistry SN2 mechanism and stereochemistry
SN2 mechanism and stereochemistry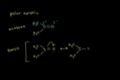 SN1 vs SN2: solvent effects
SN1 vs SN2: solvent effects SN1 vs SN2: summary
SN1 vs SN2: summary